Post-translational modifications in control of human thyroglobulin
Thyroglobulin is the protein precursor of thyroid hormones, key regulators of vertebrate development and metabolism. On the occasion of World Thyroid Day, Laura Tosatto and Francesca Coscia discuss how post-translational modifications (PTM) of thyroglobulin can regulate thyroid gland function in health and disease. They also highlight the importance of combining structural biology, proteomics, and genomics to identify PTM variations and their implications in different diseases.
The thyroid gland is an endocrine gland where thyroid hormones (THs) are synthetised and then released into the bloodstream to modulate body growth, development, and metabolism in vertebrates. Impairment of TH production in the thyroid is related to several disorders (for example Hashimoto thyroiditis, Graves’ disease, congenital hypothyroidism) which severely affect human well-being.
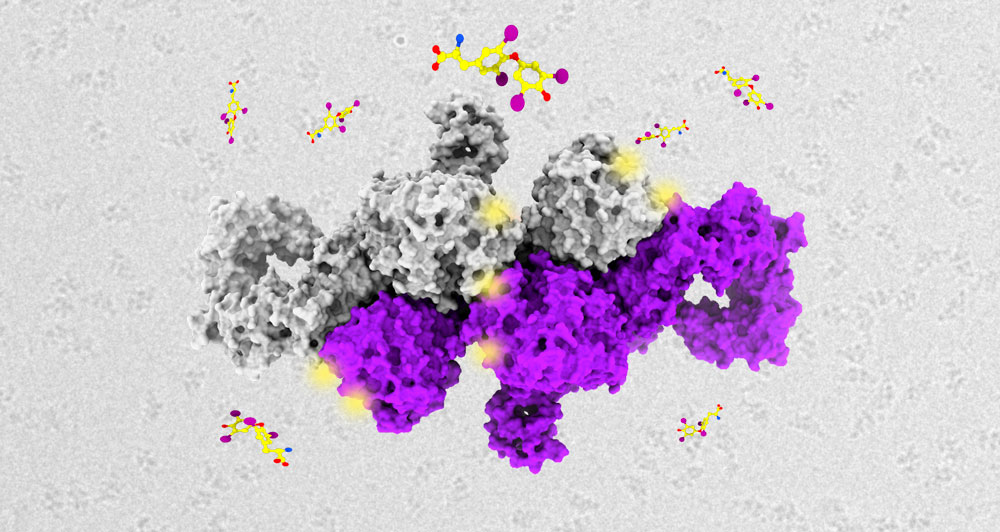
Thyroglobulin (Tg) is a high molecular mass protein produced by the follicular cells in the thyroid, which serves as an essential TH biosynthetic precursor. Thyroid function and TH production have been suggested to be regulated by a vast array of post-translational modifications (PTMs) decorating the surface of Tg (e.g. phosphorylation, glycosylation, sulfation, acetylation, and iodination).
Francesca Coscia and Laura Tosatto at the HT Structural Biology Research Centre review the different PTMs of human Tg and provide their three-dimensional representation on the recently determined electron cryo-microscopy (cryo-EM) structure of Tg (Coscia et al, Nature 2020). The researchers discuss how different PTMs may affect Tg folding and binding to other proteins, immunogenicity, and TH release. Furthermore, Tosatto and Coscia suggest that a combination of multi-omics, structural and functional data would help categorise patients’ phenotypic mutations linked to altered Tg functions and provide predictive and diagnostic tools for disease-dependent Tg PTMs patterns.
The review is published in the European Thyroid Journal.