Testa Group
The Testa Group harnesses the unprecedented potential of cell reprogramming to study the molecular basis of human neuropsychiatric and neurological diseases (NPD), by chasing the dynamics of their unfolding in physio pathologically relevant models and straddling multiple scales of analysis from single cell resolution to organismal function.
One of the most tangible outputs of somatic cell reprogramming has been a paradigm shift in our ability to model human diseases, for which fundamental limitations have been so far: i) the scarce availability of primary diseased tissues, which is particularly salient for disorders of the nervous system; and ii) the difficulty of reconstructing developmental and patient-specific trajectories during the unfolding of diseases.
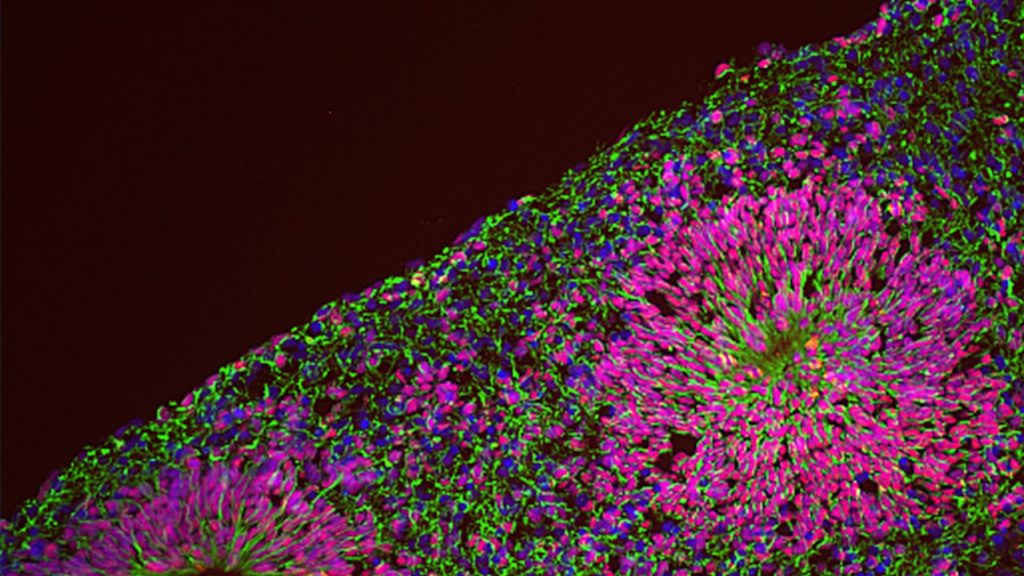
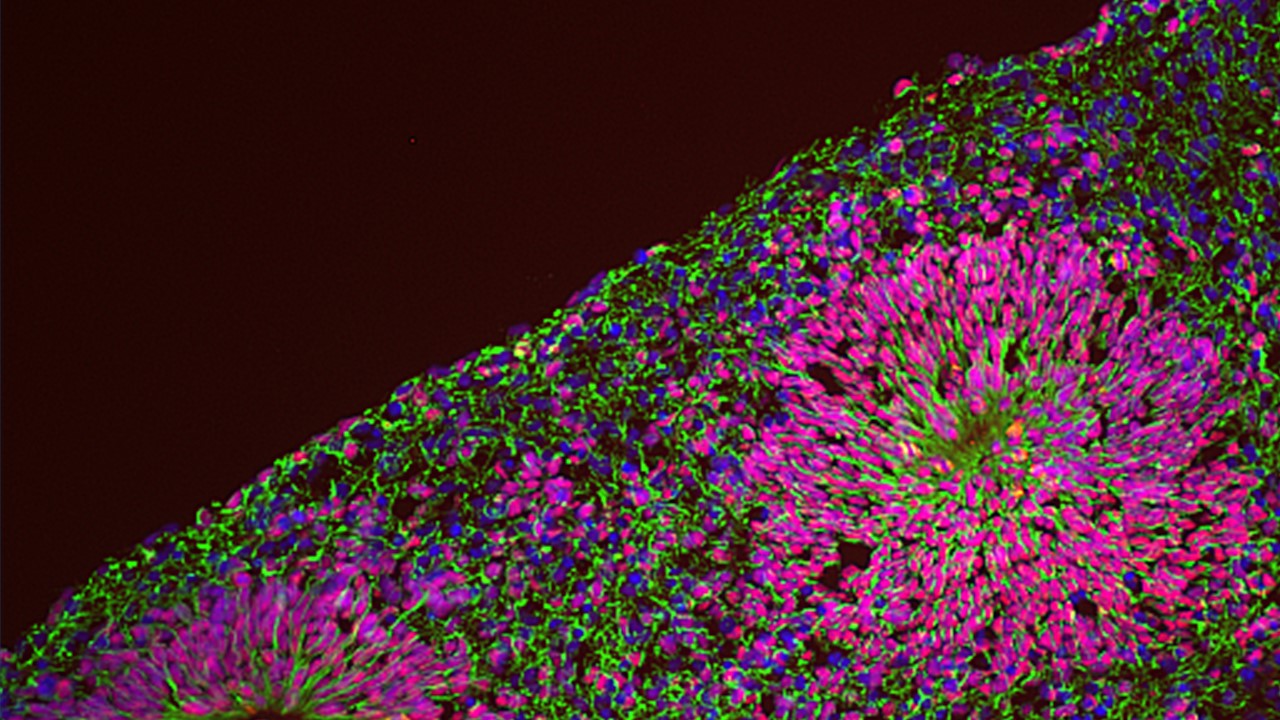
We are thus pursuing NPD modelling with human induced pluripotent stem cells (iPSC) coupled with the differentiation into relevant lineages through a range of complementary experimental paradigms, including glutamatergic neurons by induced expression of neurogenin-2 (NGN2), neural crest stem cells and 3-dimensional brain organoids that recapitulate salient stages of early brain development, including the diversity of cell populations unique to the human brain. This is allowing us to dissect the genetic and environmental components of NPD pathogenesis, by means of several “omics” approaches at a bulk and single cell resolution integrated with high throughput imaging and functional in vitro and in vivo assays.
The Testa Group focuses on a uniquely informative range of syndromes featuring intellectual disability and autism spectrum disorder that are caused by mutations or dosage alterations in epigenetic regulators and transcription factors, including Williams-Beuren Syndrome and 7q11.23 microduplication Syndrome, Kabuki Syndrome, ADNP-related autistic spectrum disorders, Weaver Syndrome, Gabriele-Testa-DeVries Syndrome, as well as on paradigmatic environmental factors that adversely impact on neurodevelopment, namely endocrine disruptive chemicals.
Finally, the prism of human neurodevelopmental disorders is also allowing us to investigate the logic of the gene regulatory networks underlying the evolution of the modern human face and brain, integrating the analysis of craniofacial dysmorphologies and brain alterations to illuminate the evolutionary-developmental trajectories underlying the modern human condition.
Testa Group 2023

Group members
-
 Giuseppe Testa
Giuseppe Testa
Head of Neurogenomics -
 Davide Aprile
Davide Aprile
Postdoc -
 Lorenzo Basile
Lorenzo Basile
Undergraduate Intern -
 Nicolò Caporale
Nicolò Caporale
Postdoctoral Associate -
 Davide Castaldi
Davide Castaldi
Postdoc Fellow -
 Lorenza Culotta
Lorenza Culotta
Senior Technician -
 Francesco Elli
Francesco Elli
Affiliate Scientist -
 Marlene Cristina Faria Pereira
Marlene Cristina Faria Pereira
Affiliate Scientist -
 Samuele Galimi
Samuele Galimi
Postgraduate Fellow -
 Oliviero Leonardi
Oliviero Leonardi
PhD Student -
 Lisa Mainardi
Lisa Mainardi
Scientific Visitor -
 Alessandro Melon
Alessandro Melon
Undergraduate Intern -
 Riccardo Nagni
Riccardo Nagni
Undergraduate Intern -
 Marco Tullio Rigoli
Marco Tullio Rigoli
Affiliate Scientist -
 Ludovico Rizzuti
Ludovico Rizzuti
Affiliate Scientist -
 Adrianos Skaros
Adrianos Skaros
Affiliate Scientist -
 Sarah Stucchi
Sarah Stucchi
PhD Student -
 Alejandro Lopez Tobon
Alejandro Lopez Tobon
Scientific Advisor -
 Alessandro Valente
Alessandro Valente
Affiliate Scientist -
 Alessia Valenti
Alessia Valenti
PhD Student -
 Emanuele Villa
Emanuele Villa
Senior Staff Scientist -
 Alessandro Vitriolo
Alessandro Vitriolo
Affiliate Scientist -
 Narges Yahyazadeh
Narges Yahyazadeh
Scientific Visitor
Publications
-
07/2021 - Autophagy
Imbalanced autophagy causes synaptic deficits in a human model for neurodevelopmental disorders
Macroautophagy (hereafter referred to as autophagy) is a finely tuned process of programmed degradation and recycling of proteins and cellular components, which is crucial in neuronal function and synaptic integrity. Mounting evidence implicates chromatin remodeling in fine-tuning autophagy pathways. However, this epigenetic regulation is poorly understood in neurons. Here, we investigate the role in autophagy […]
-
12/2020 - The EMBO Journal
COVID-19 lessons from the dish: Dissecting CNS manifestations through brain organoids
COVID-19 is increasingly understood as a systemic disease with pathogenic manifestations beyond the respiratory tract. Recent work by Ramani et al (2020) dissects the cellular and molecular mechanisms of SARS-CoV-2’s neurotrophic properties, using viral exposure of human brain organoids. Their findings highlight neurons as primary target of cerebral SARS-CoV-2 infection and uncover its Tau-related neurotoxicity.
-
11/2020 - Molecular Autism
High-throughput screening identifies histone deacetylase inhibitors that modulate GTF2I expression in 7q11.23 microduplication autism spectrum disorder patient-derived cortical neurons
Background Autism spectrum disorder (ASD) is a highly prevalent neurodevelopmental condition affecting almost 1% of children, and represents a major unmet medical need with no effective drug treatment available. Duplication at 7q11.23 (7Dup), encompassing 26–28 genes, is one of the best characterized ASD-causing copy number variations and offers unique translational opportunities, because the hemideletion of […]
-
09/2020 - Molecular Autism
Autism spectrum disorder at the crossroad between genes and environment: contributions, convergences, and interactions in ASD developmental pathophysiology
The complex pathophysiology of autism spectrum disorder encompasses interactions between genetic and environmental factors. On the one hand, hundreds of genes, converging at the functional level on selective biological domains such as epigenetic regulation and synaptic function, have been identified to be either causative or risk factors of autism. On the other hand, exposure to […]
-
09/2020 - Nature
LifeTime and improving European healthcare through cell-based interceptive medicine
LifeTime aims to track, understand and target human cells during the onset and progression of complex diseases and their response to therapy at single-cell resolution. This mission will be implemented through the development and integration of single-cell multi-omics and imaging, artificial intelligence and patient-derived experimental disease models during progression from health to disease. Analysis of […]